Difference between revisions of "Contrib/CompressibleMixingPhaseChangeFoam"
Mkraposhin (Talk | contribs) (→Model Equations) |
Mkraposhin (Talk | contribs) |
||
| Line 17: | Line 17: | ||
where <math>\hat \rho</math> computed with respect to previous formulations | where <math>\hat \rho</math> computed with respect to previous formulations | ||
| − | |||
mixture density <math> \rho </math> calculated as | mixture density <math> \rho </math> calculated as | ||
| Line 26: | Line 25: | ||
</math> | </math> | ||
| + | Here, and afterthere indices: | ||
| + | |||
| + | 1,l,f - for liquid (heavy media with low compressibility) | ||
| + | |||
| + | 2,g,s - for gas (light media (like steam) with big compressibility) | ||
| + | |||
| + | without index - mixture variable (or all variables local to some phase) | ||
* Liquid volume transport | * Liquid volume transport | ||
| Line 79: | Line 85: | ||
+ | + | ||
\nabla \cdot \rho \textbf{U} \textbf{U} = \nabla \cdot R^{Eff} - \nabla p + \rho \textbf{g} | \nabla \cdot \rho \textbf{U} \textbf{U} = \nabla \cdot R^{Eff} - \nabla p + \rho \textbf{g} | ||
| + | </math> | ||
| + | by substituting piezometric pressure <math> \hat p = p - \rho \textbf{g} \textbf{x} </math> | ||
| + | we get: | ||
| + | <math> | ||
| + | \frac {\partial \rho \textbf{U}}{\partial t} | ||
| + | + | ||
| + | \nabla \cdot \rho \textbf{U} \textbf{U} = \nabla \cdot R^{Eff} - \nabla \hat p - \textbf{g} \cdot \nabla \rho | ||
</math> | </math> | ||
| Line 91: | Line 104: | ||
</math> | </math> | ||
| − | |||
* Energy equation | * Energy equation | ||
| + | Energy equation for mixture temperature obtained from sum of energy equations for each phase. Consider energy equation for phase-1: | ||
| + | |||
| + | <math> | ||
| + | \frac{\partial \alpha_1 \rho_1 e_1} {\partial t} + | ||
| + | \nabla \cdot \left ( \alpha_1 \rho_1 e_1 \textbf{U} \right ) - \nabla \cdot \textbf{q}_1 | ||
| + | = | ||
| + | \dot m_1 e_1 | ||
| + | </math> | ||
| + | |||
| + | by converting to enthalpies we get: | ||
| + | |||
| + | <math> | ||
| + | \frac{\partial \alpha_1 \rho_1 h_1} {\partial t} + | ||
| + | \nabla \cdot \left ( \alpha_1 \rho_1 h_1 \textbf{U} \right )- | ||
| + | \left ( | ||
| + | \frac {\partial \alpha_1 p}{\partial t} + \nabla \cdot \left ( \alpha_1 p \textbf{U} \right ) | ||
| + | \right ) | ||
| + | - \nabla \cdot \textbf{q}_1 | ||
| + | = | ||
| + | \dot m_1 h_1 - \dot m_1 \frac{p}{\rho_1} | ||
| + | </math> | ||
| + | |||
| + | By substituting temperature instead of enthalpy, after conversion to volume fluxes we get equation for temperature (divided by | ||
| + | <math>\rho_1 C_{p,1} </math> ) | ||
| + | |||
| + | <math> | ||
| + | \alpha_1 \left (\frac{\partial T}{\partial t} + \nabla \cdot \left ( T \textbf{U} \right ) \right ) | ||
| + | + T \frac{\alpha_1}{\rho_1} \frac{d \rho_1}{d t} + T \frac{d \alpha_1}{d t} - \frac{1}{\rho_1 C_{p,1}} \nabla \cdot \textbf{q}_1 | ||
| + | = | ||
| + | T \frac{\dot m_1}{\rho_1} - \frac{1}{\rho_1 C_{p,1}} \frac{p}{\rho_1} \dot m_1 + | ||
| + | \frac{1}{\rho_1 C_{p,1}} | ||
| + | \left ( | ||
| + | \frac {\partial \alpha_1 p}{\partial t} + \nabla \cdot \left ( \alpha_1 p \textbf{U} \right ) | ||
| + | \right ) | ||
| + | </math> | ||
| + | |||
| + | By combining equations of phases, we get energy balance for mixture: | ||
| + | |||
| + | <math> | ||
| + | \frac{\partial T}{\partial t} + \nabla \cdot \left ( T \textbf{U} \right ) - T \nabla \cdot \textbf{U} | ||
| + | -\frac{1}{\rho_1 C_{p,1}} \nabla \cdot \kappa_1 \nabla T -\frac{1}{\rho_2 C_{p,2}} \nabla \cdot \kappa_2 \nabla T | ||
| + | = | ||
| + | \left ( \frac{\alpha_1}{\rho_1 C_{p,1}} + \frac{\alpha_2}{\rho_2 C_{p,2}} \right ) \frac{d p}{d t} | ||
| + | + \frac{p}{\rho_1 C_{p,1}}\frac{d \alpha_1}{d t} + \frac{p}{\rho_2 C_{p,2}}\frac{d \alpha_2}{d t} | ||
| + | + \dot m_1 p \left (-\frac{1}{\rho_1 \rho_1 C_{p,1}} + \frac{1}{\rho_2 \rho_2 C_{p,2}}\right ) | ||
| + | </math> | ||
| + | |||
| + | * Phase change model | ||
[http://www.os-cfd.ru/compressibleMixingPhaseChangeFoam/Solver.tgz Solver sources and tutorials located here] | [http://www.os-cfd.ru/compressibleMixingPhaseChangeFoam/Solver.tgz Solver sources and tutorials located here] | ||
Revision as of 17:06, 30 December 2012
Solver for two fluids with phase change (for example - water <---> steam), pressure and temperature density dependence
Model Equations
- Equation of state
Low-compressible fluid: 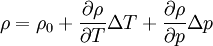
Ideal gas: 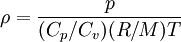
By combining this equations, we can get general relation:
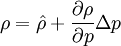
where 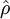 computed with respect to previous formulations
computed with respect to previous formulations
mixture density 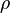 calculated as
calculated as
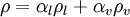
Here, and afterthere indices:
1,l,f - for liquid (heavy media with low compressibility)
2,g,s - for gas (light media (like steam) with big compressibility)
without index - mixture variable (or all variables local to some phase)
- Liquid volume transport
Let us consider transport of liquid (heavy phase) volume fraction 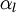 :
:
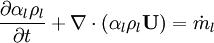
By converting to volume fluxes we get:
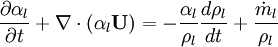
Using equation of state, we can reformulate substantial derivative for density in terms of pressure for any phase:
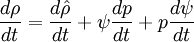
- General rule for converting from mass to volume fluxes in transport equation
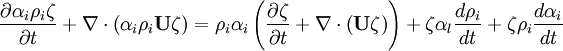
- Momentum equation (velocity prediction)
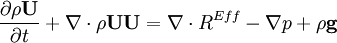 by substituting piezometric pressure
by substituting piezometric pressure 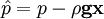 we get:
we get:
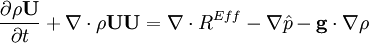
- Pressure equation obtained by summation of equation for volume phase fraction of liquid and gas phases:
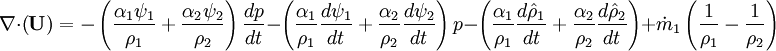
- Energy equation
Energy equation for mixture temperature obtained from sum of energy equations for each phase. Consider energy equation for phase-1:
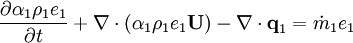
by converting to enthalpies we get:
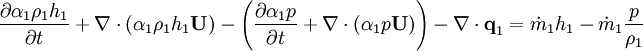
By substituting temperature instead of enthalpy, after conversion to volume fluxes we get equation for temperature (divided by
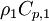 )
)
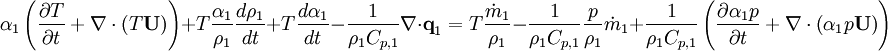
By combining equations of phases, we get energy balance for mixture:
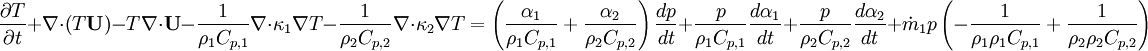
- Phase change model